DATA
We have developed a family of monoclonal antibodies that have the same specificity based on a binding, complementarity determining region (“CDR”) sequence homology. Our lead product is a humanized monoclonal antibody (our “mAb” or “SIWA 318H”). It targets a SIWA-identified, naturally-occurring cell surface marker (our “SIWA Marker”) on both cancer cells and senescent cells (“SCs”), which cells are thereby destroyed and removed through normal processes. Our family of antibodies includes SIWA 318M, a murine homolog that we used in our early in vitro and in vivo studies.
SIWA has conducted several in vivo preclinical studies. which have shown that using SIWA 318 over a short period of time has been able to statistically significantly:
- Reduce senescent cells as measured by p16INK4a
- Increase muscle mass in a naturally aged mouse model
- Inhibit metastatic lung foci in a 4T1 breast cancer model
Across these studies, no adverse effects from SIWA 318 were recorded.
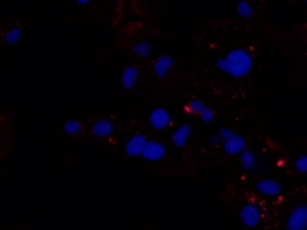
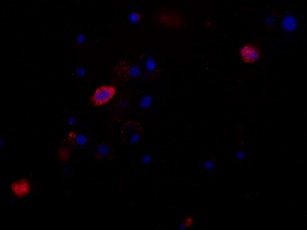
318H In Vitro Binding Results
318H Binding to PANC-1 cells - Immunoflourescence


318H Binding to Orthotopic PSN1 in Mouse – Immunohistochemistry

318H binding to Patient Derived Pancreatic Cancer cells – immunohistochemistry


318H binding to MCF-7 cells (Breast cancer) - Immunofluorescence


318H ─ Binding to HTB-14 Glioblastoma cells and nuclei from an ATCC glioma cell line panel
318H In Vitro Binding Results
Design of the Proof-of-concept (POC)/ efficacy study for 318H in humanized mice for treatment of subcutaneous pancreatic cancer

318H treatment reduced pancreatic tumor growth in humanized mice model

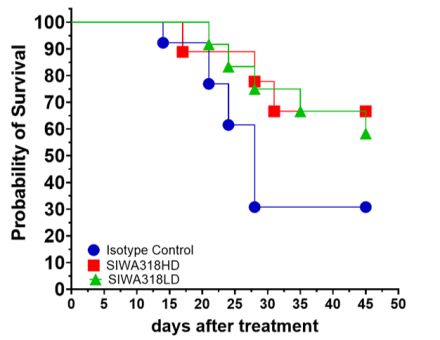
Increased Survival in humanized mice treated with 318H mAb
Complete remission* response in humanized mice treated with 318H
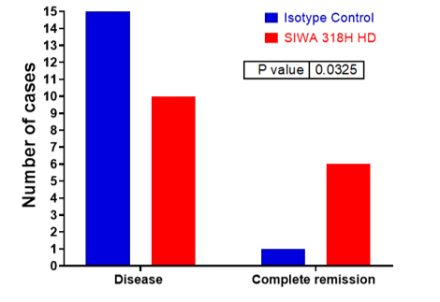
| Isotype Control | SIWA 318H HD | |
|---|---|---|
| Disease | 15 | 10 |
| Complete Remission | 1 | 6 |

| Isotype Control | SIWA 318H LD | |
|---|---|---|
| Disease | 15 | 9 |
| Complete Remission | 1 | 7 |
*Complete remission is defined as the disappearance of all signs of tumor presence in response to treatment
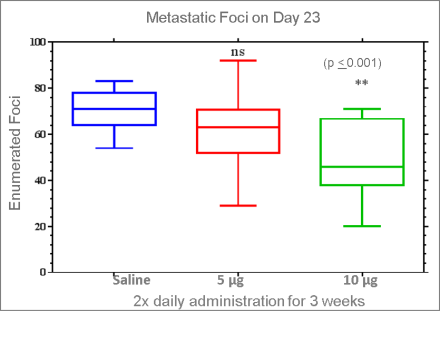
Efficacy In A Triple Negative Breast Cancer Metastasis Model
Design of the murine model for the treatment of metastasis in breast with 318M (mouse homolog)

318M (318H Mouse Homolog) Reduced metastasis in breast cancer model
Some Other Images of 318H Binding Results